Latest News
AYUSH Asks Patanjali To Refrain From Advertising Its COVID-19 Cure
The Ministry of AYUSH has asked Patanjali Ayurved Ltd. To refrain from advertising its medicine for covid-19 until it gets verified. While taking suo moto cognizance of the media report, the ministry said that facts of the claim and details of the medicine are not known and uncorroborated.
The ministry has asked the company not to make any claims until the treatment gets duly verified by the concerned authority. The ministry has further asked the State Licensing Authority of Uttrakhand Government to provide the copy of company’s license and details of the product approval of the Ayurvedic medicines that the company claims is the treatment of covid-19.
It was further informed that the advertisement of Ayurvedic and other medicines are governed under Drugs and Magic Remedies (Objectionable Advertisements) Act, 1954. The ministry also issued a notice on April 21, 2020 informing about all the perquisites and manner in which the research of covid-19 treatment has to be taken place.
For verification, the ministry has asked the company to provide:
- The name and composition of the treatment.
- Place (site/hospital) where the research was done.
- Protocol, sample size, Institutional Ethics Committee clearance, CTRI registration and results data of the study(ies).
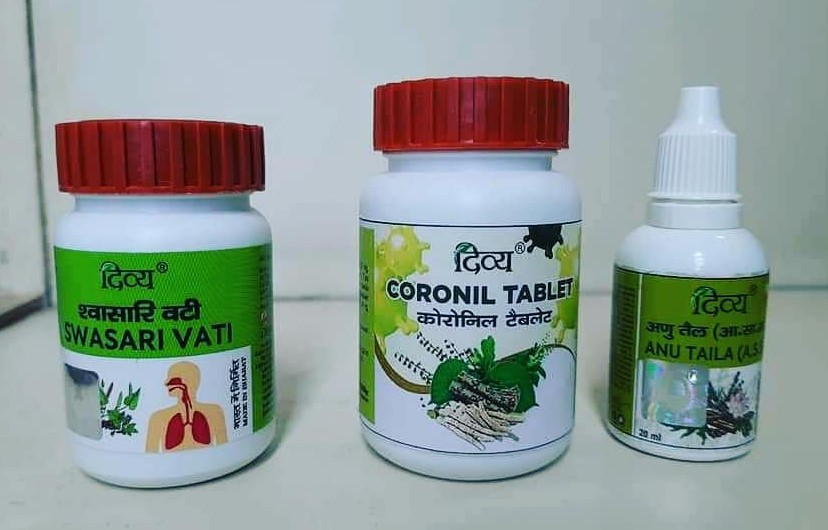
Before any verification, the company has launched 'Coronil and Swasari' and is claimed to show "100% favourable results" during clinical trials. Yog Guru Baba Ramdev said in the Press Conference "We are launching COVID medicines Coronil and Swasari today. We conducted two trials of these, first clinical controlled study, which took place in Delhi, Ahmedabad, among many other cities. Under this 280 patients were included and 100 per cent of those recovered. We were able to control Corona and its complications in this. After this the all-important clinical control trial was conducted,"
https://www.livelaw.in/news-updates/centre-asks-patanjali-to-stop-advertisements-about-unverified-ayurveda-medicine-developed-to-cure-covid-19-158786?infinitescroll=1
https://www.livelaw.in/news-updates/centre-asks-patanjali-to-stop-advertisements-about-unverified-ayurveda-medicine-developed-to-cure-covid-19-158786?infinitescroll=1